

Telephone: +44 (0)1252 405186
Email: tsc@gordonengland.co.uk
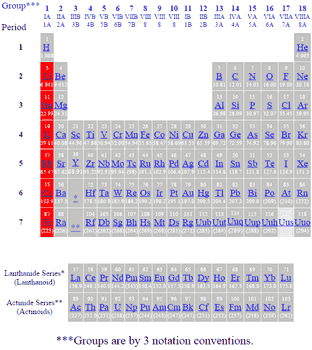
The alkali metals are a series of elements comprising: lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Note that hydrogen, although nominally placed in the same group, rarely exhibits behavior comparable to the alkali metals (Under extremely high pressure, hydrogen can become metallic and then behaves like an alkali metal).
The alkali metals are all highly reactive and tend not to be found in elemental form in nature.
Alkali metals have the lowest ionisation energy in their respective periods, as removing the single electron from the outermost shell gives them the stable noble gas configuration. But their second ionisation potentials are very high, as removing an electron from a species having a noble gas configuration is very difficult.
| Group*** Period | |||||||||||||||||||
| 1 IA 1A | 2 IIA 2A | 3 IIIB 3B | 4 IVB 4B | 5 VB 5B | 6 VIB 6B | 7 VIIB 7B | 8 VIII 8 | 9 VIII 8 | 10 VIII 8 | 11 IB 1B | 12 IIB 2B | 13 IIIA 3A | 14 IVA 4A | 15 VA 5A | 16 VIA 6A | 17 VIIA 7A | 18 VIIIA 8A | ||
| 1 | 1 H 1.008 | 2 He 4.003 | |||||||||||||||||
| 2 | 3 Li 6.941 | 4 Be 9.012 | 5 B 10.81 | 6 C 12.01 | 7 N 14.01 | 8 O 16.00 | 9 F 19.00 | 10 Ne 20.18 | |||||||||||
| 3 | 11 Na 22.99 | 12 Mg 24.31 | 13 Al 26.98 | 14 Si 28.09 | 15 P 30.97 | 16 S 32.07 | 17 Cl 35.45 | 18 Ar 39.95 | |||||||||||
| 4 | 19 K 39.10 | 20 Ca 40.08 | 21 Sc 44.96 | 22 Ti 47.88 | 23 V 50.94 | 24 | 25 | 26 Fe 55.85 | 27 Co 58.47 | 28 Ni 58.69 | 29 | 30 Zn 65.39 | 31 Ga 69.72 | 32 Ge 72.59 | 33 As 74.92 | 34 Se 78.96 | 35 Br 79.90 | 36 Kr 83.80 | |
| 5 | 37 Rb 85.47 | 38 Sr 87.62 | 39 Y 88.91 | 40 Zr 91.22 | 41 Nb 92.91 | 42 Mo 95.94 | 43 Tc (98) | 44 Ru 101.1 | 45 Rh 102.9 | 46 Pd 106.4 | 47 Ag 107.9 | 48 Cd 112.4 | 49 In 114.8 | 50 Sn 118.7 | 51 Sb 121.8 | 52 Te 127.6 | 53 I 126.9 | 54 Xe 131.3 | |
| 6 | 55 Cs 132.9 | 56 Ba 137.3 | * | 72 Hf 178.5 | 73 Ta 180.9 | 74 W 183.9 | 75 Re 186.2 | 76 Os 190.2 | 77 Ir 190.2 | 78 Pt 195.1 | 79 Au 197.0 | 80 Hg 200.5 | 81 Tl 204.4 | 82 Pb 207.2 | 83 Bi 209.0 | 84 Po (209) | 85 At (210) | 86 Rn (222) | |
| 7 | 87 Fr (223) | 88 Ra (226) | ** | 104 Rf (261) | 105 Db (262) | 106 Sg (266) | 107 Bh (264) | 108 Hs (269) | 109 Mt (268) | 110 Ds (281) | 111 Rg (272) | 112 Cn (285) | 113 Nh (284) | 114 Fl (289) | 115 Mc (288) | 116 Lv (292) | 117 Ts (294) | 118 Og (294) | |
|
| |||||||||||||||||||
| Lanthanide Series* (Lanthanoid) | 57 La 138.9 | 58 Ce 140.1 | 59 Pr 140.9 | 60 Nd 144.2 | 61 Pm (145) | 62 Sm 150.4 | 63 Eu 152.0 | 64 Gd 157.3 | 65 Tb 158.9 | 66 Dy 162.5 | 67 Ho 164.9 | 68 Er 167.3 | 69 Tm 168.9 | 70 Yb 173.0 | 71 Lu 175.0 | ||||
| Actinide Series** (Actinoids) | 89 Ac (227) | 90 Th 232.0 | 91 Pa (231) | 92 U (238) | 93 Np (237) | 94 Pu (244) | 95 Am (243) | 96 Cm (247) | 97 Bk (247) | 98 Cf (251) | 99 Es (252) | 100 Fm (257) | 101 Md (258) | 102 No (259) | 103 Lr (262) | ||||


Introducing
Nature of Thermal Spray Coatings
Surface Engineering in a Nutshell
Surface Engineering Forum
Thermal Spray Gun Repair Service
Plasma Consumable Parts
Thermal Spray Powder Supplies
Applications:
Thermal Spray Coatings on Carbon and Glass Fibre Reinforced Polymers
HVOF Coating of Paper Making Roll
Abradable Coatings
Thermal Spray Processes:
Combustion Wire Thermal Spray Process
Combustion Powder Thermal Spray Process
Arc Wire Thermal Spray Process
Plasma Thermal Spray Process
HVOF Thermal Spray Process
HVAF Thermal Spray Process
Detonation Thermal Spray Process
Plasma Flame Theory
Cold Spray Coating Process
Wear and Use of Thermal Spray Coatings
Corrosion and Use of Thermal Spray Coatings
Glossary of Thermal Spray and Surface Engineering Terms
Image Directory for Thermal Spray Coatings
Plasma Gas Flow Information
Plasma Gas Flow Correction Calculator
Contact Form
Links to other interesting sites related to thermal spray and surface engineering
Reciprocal Links
Periodic Table of the Elements
SI Units
Calculators for Conversion between Units of Measurement
Hardness Testing
Surface Engineering Message Board Archive
Surface Engineering Message Board Archive Index
Photography Gallery2
Photography Gallery3
© Copyright Gordon England